Scientific confidence: High

In the absolute darkness between one and three kilometers down, where pressure exceeds two hundred atmospheres and cold hovers near two degrees Celsius, a female abyssal anglerfish — likely *Melanocetus* or a close relative — holds herself suspended in open midwater with barely a flicker of her caudal fin, her entire metabolic existence calibrated for patience and minimal energy expenditure. The only light in this volume of ocean is her own: the esca, a modified dorsal spine tipped with a symbiotic colony of bioluminescent bacteria, pulses with a steady blue-green glow that is simultaneously a fishing lure, a private language, and a weapon — drawing prey organisms upward through the marine snow, their small bodies betrayed by the very curiosity that evolution built into them. Each drifting particle of marine snow — fragmented fecal pellets, shed mucus, dead zooplankton descending from the sunlit world far above — passes briefly through that living light, then vanishes back into blackness, a quiet rain that constitutes much of the energy budget for this entire zone. Her recurved teeth, translucent as glass and needle-thin, are evolved not to cut but to cage: anything that enters that crescent jaw will find no mechanical escape, the recurved geometry ensuring closure is irreversible. Here the ocean exists in a state that preceded the first eye and will persist long after the last one closes — pressurized, lightless except for its own cold fire, and entirely indifferent to being known.

Between 1,500 and 2,500 meters, no photon from the sun has ever reached, and the water pressing inward from all directions exceeds 200 atmospheres — a crushing stillness in which warm-blooded intuitions about the ocean dissolve entirely. Here, Chauliodus sloani, the viperfish, is one of the midnight zone's most ruthless ambush predators: its elongated dorsal ray carries a bioluminescent tip used as a lure, while the lower fangs, too long to retract, lock upward like paired daggers when the jaws hinge open at extraordinary gape angles, allowing it to engulf prey approaching its own body length. The intermittent cyan-blue pulses threading through the disturbed cloud of deep pelagic crustaceans — likely euphausiids and hyperiid amphipods — are not coincidental decoration; they are alarm bioluminescence, a last chemical scream propagating outward through the swarm as bodies collide and scatter in three-dimensional panic, each flash briefly silhouetting the viperfish's gunmetal flanks and reflective tapetal eyes before the darkness reseals itself. Fine marine snow drifts indifferently through the entire corridor, organic particulate descending from the productive layers far above, the slow rain of carbon that feeds this entire dark ecosystem and connects the sunlit surface to a world that requires no light to function. The silence here is not empty — it is structured, pressurized, and teeming with predation invisible to every wavelength but its own.

In the absolute darkness between one and four kilometers below the surface, where pressure exceeds two hundred atmospheres and sunlight has been extinct for hundreds of meters above, a black dragonfish — most likely a species of *Idiacanthus* or a relative within the family Stomiidae — hangs motionless in open midwater, its elongate body so deeply melanized that it absorbs nearly all bioluminescent photons that strike it, rendering it a near-perfect void within a void. Along its flank, rows of cyan photophores — intrinsic light organs derived from modified scales and underlying chromatophores — emit a species-specific signal pattern, a private wavelength tuned to conspecific eyes and largely invisible to prey with narrower spectral sensitivity; the slender chin barbel, trailing below the jaw like a filament of living glass, serves simultaneously as a lure and a sensory probe in water that carries no current strong enough to disturb it. Marine snow — the constant slow precipitation of organic detritus, fecal pellets, and bacterial aggregates that constitutes the primary energy transfer from the sunlit surface to these depths — drifts through the faint bioluminescent glow in all directions, each particle a slow-falling reminder that this darkness is not empty but metabolically connected to the world above. The surrounding water column holds no floor and no ceiling within any perceptible distance, only an expanding volume of cold, clear, pressurized ocean where every organism that passes must carry its own light or live entirely without it — a world of intermittent, purposeful glimmers suspended in geological silence, existing on its own terms across timescales that have nothing to do with human witness.

At the upper margin of the midnight zone, a goblin shark (*Mitsukurina owstoni*) drifts in near-total darkness above a fractured basalt escarpment, its elongated rostrum and pallid, pressure-softened body suspended between an almost imperceptible remnant of deep blue descending from hundreds of meters above and the absolute black of the water column below. Here, at pressures approaching one hundred atmospheres, sunlight has long since ceased to exist as a meaningful quantity, leaving only bioluminescence — scattered cyan and blue-green pinpoints from unseen planktonic organisms drifting like cold embers through the dark — as the only illumination this world knows. The goblin shark's flabby, lightly mineralized body, adapted to the chemical and mechanical demands of the deep, hangs poised and slack in that silent way unique to predators that do not need to rush, their sensory systems finely tuned to detect the faintest pressure wave or electrochemical trace in cold, 3–4 °C water. Below, the basalt escarpment falls away in fractured volcanic ledges dusted with sparse mineral crusts, shaped by geological forces indifferent to the living world that slowly colonizes their shadows. Marine snow descends in all directions through the immense empty water column, tracing the invisible slow pulse of an ocean that has existed in this darkness, under this pressure, in this profound silence, entirely without witness.

In the permanent darkness between one and four kilometers down, where pressure exceeds two hundred atmospheres and cold hovers near three degrees Celsius, a Magnapinna squid hangs motionless in the water column — its broad triangular fins and semi-transparent mantle barely materializing against an ocean that is essentially infinite black. This genus, among the least understood cephalopods on Earth, is distinguished by its extraordinary arm-filaments, which extend far beyond the body length and bend at distinctive elbow-like angles before trailing downward into the void; current hypotheses suggest these filaments may passively intercept small prey drifting through the marine snow corridor, a strategy suited to a world where metabolic economy is survival. Scattered cyan and blue bioluminescent pulses from distant plankton drift through the water column like cold sparks, their living light the only illumination in a zone where photosynthetic radiation expired hundreds of meters above, and fine particulates of marine snow — the slow rain of organic matter from shallower ecosystems — drift past the squid's tissues with no urgency whatsoever. Far behind and below, an almost imperceptible warm tint bleeds through the water, the faintest atmospheric signature of hydrothermal activity on a distant ridge, chemiluminescent rather than thermal at this remove, coloring nothing but hinting at the geological restlessness underlying even this stillness. This is the ocean as it has always been: pressurized, cold, biologically precise, and entirely indifferent to any witness.
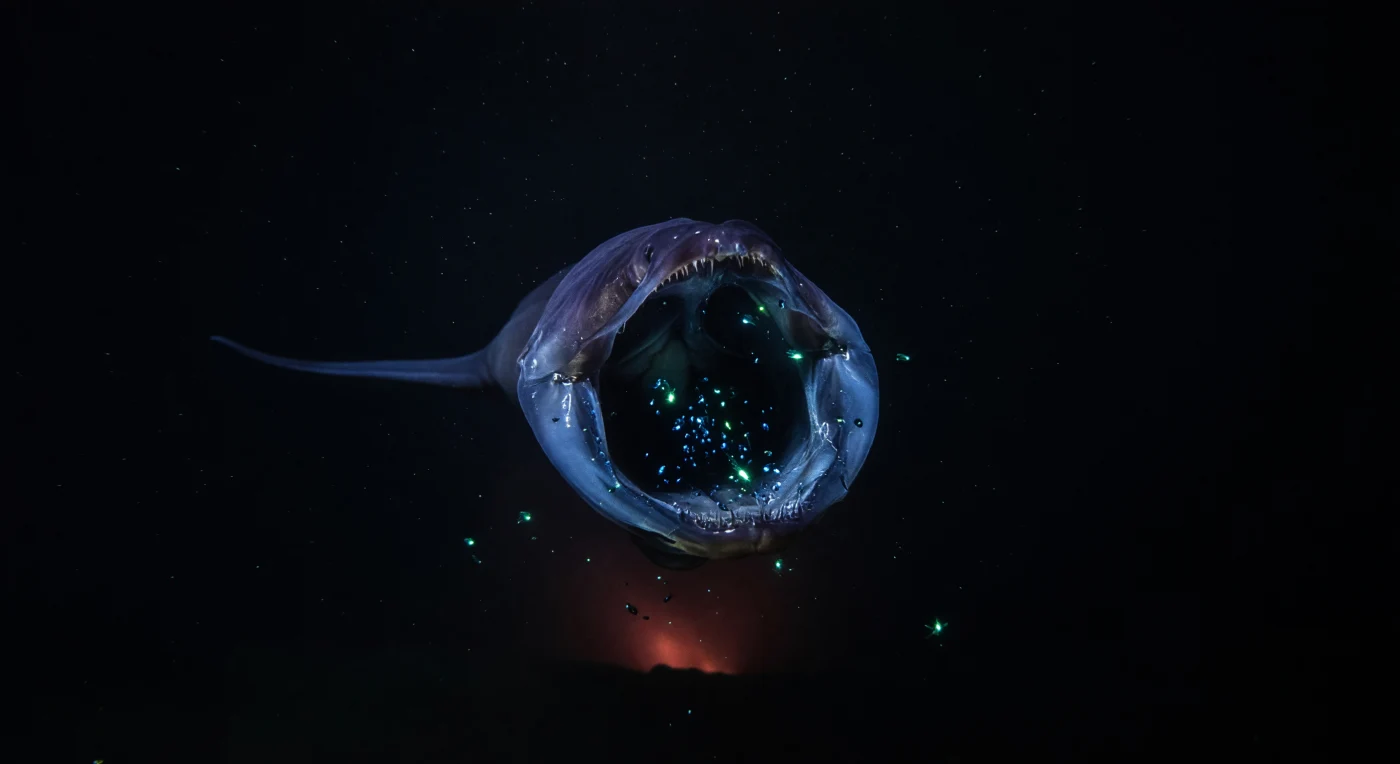
At depths where pressure exceeds two hundred atmospheres and the last photon of sunlight died kilometers above, a gulper eel — Eurypharynx pelecanoides — executes one of the ocean's most extreme feeding strategies: its loosely hinged jaw, unmoored from rigid skeletal constraint, balloons outward into a translucent bluish membrane large enough to engulf prey several times the eel's own girth, the distended oral cavity briefly lit from within by the panicked bioluminescent pulses of trapped organisms whose cold cyan flickers silhouette needle-fine teeth and slick, stretched tissue. The eel's body, black-violet and gelatinous under pressure that would collapse any gas-filled space, tapers to a whip-thin tail threading back into absolute darkness, while sparse marine snow — the slow organic rain of decomposing matter that constitutes the primary nutrient corridor between the sunlit surface and this lightless realm — drifts freely past in every direction, indifferent to the violence occurring within it. Far below, a barely perceptible orange-red thermal tint in the deepest part of the water column hints at hydrothermal chemistry, a reminder that this zone is geologically as well as biologically dynamic, structured by mid-ocean ridges, seamount flanks, and the chemical plumes they release. Here, at two to three thousand meters, predation is not a daily rhythm but an event of rare consequence, shaped entirely by bioluminescent deception, sensory acuity, and anatomy evolved over millions of years in a world that has never required the witness of any other.

Along a mid-ocean ridge fracture somewhere between one and four kilometers beneath the surface, freshly emplaced basalt bleeds dim red-orange heat through its cracked glassy crust, the only thermal light in an otherwise lightless world, while hydrothermal plumes rise in soft chemiluminescent veils through near-freezing water pressing down at hundreds of atmospheres. An anglerfish moves with glacial patience past fractured lava ledges, its bioluminescent lure pulsing blue-white ahead of a gape of translucent recurved teeth, while bioluminescent plankton brushed aside by its passage flicker briefly cyan and green against its flanks — a momentary betrayal of motion in a world that rewards stillness. Deeper in the composition, a viperfish hangs in near-perfect suspension, its needle fangs and dark metallic skin catching the faint microbial sheen rising from mineralized surfaces, and farther back in the column the Magnapinna squid trails its impossibly elongated arm filaments downward like pale ribbons dissolving into black. This is a world of patchy food and lethal patience, where evolution has refined stealth, sensory amplification, and bioluminescent deception across millions of years of unwitnessed darkness — a predator guild shaped entirely by pressure, cold, and the utter absence of light that did not first arise within a living body.

At depths where pressure exceeds two hundred atmospheres and the last trace of solar radiation has long since been extinguished, a sulfide-mineral landscape rises from fresh basalt in towering chimneys that discharge superheated fluids instantly quenched into billowing black plumes — a chemistry of iron, manganese, and sulfide compounds precipitating in real time against water hovering near two degrees Celsius. Along the plume margin, a bathypelagic predator moves with the conserved precision of an animal that cannot afford wasted motion: its musculature is soft and pressure-tolerant, its sensory systems finely tuned to the mechanical signatures and faint bioluminescent sparks that constitute the only available information in this absolute dark. Scattered through the surrounding water column, the scattered blue-green points of bioluminescence — produced by photophores, secreted luciferin compounds, and symbiotic bacteria across dozens of organisms — represent not decoration but a language of lures, counter-illumination, and predator-prey signaling refined across millions of years of evolution beneath any sunlight. Deeper in the darkness, the ghostly suggestion of a Magnapinna squid extends arms of improbable length into water laden with mineral snow and organic detritus drifting down from the productive surface far above, while the faint rust-orange chemiluminescent shimmer of the hottest vent apertures writes the only warm color into a world otherwise composed entirely of black, cold, and the cold fire of living light.

At the edge of a diffuse hydrothermal seep along a mid-ocean ridge, where mineral-laden warm water bleeds upward through fractured basalt at roughly 200 to 300 atmospheres of pressure, a small anglerfish holds its ambush position with absolute stillness in water near 2 °C. Its esca — the bioluminescent lure evolved through millions of years of selection in perpetual darkness — casts the only concentrated light in the scene, a blue-green point generated by symbiotic bacteria housed in specialized tissue, while sparse chemiluminescent pulses from vent-associated fauna and microbial mats add faint cyan halos across sulfur-pale mineral crusts and glassy basalt. Marine snow, the slow rain of organic particulates descending from the sunlit ocean more than a kilometer above, drifts freely through the water column, each flake a fragment of the biological pump connecting this lightless world to the surface biosphere. The thermal boundary where warm vent effluent meets near-freezing deep seawater creates a refractive shimmer — a chemocline visible in the slight distortion of background dark — concentrating prey drawn to elevated nutrients and microbial productivity at this narrow, predator-rich margin. Here, without witness, without season, the ocean's oldest predatory logic plays out in cold silence: patience, a single glowing lure, jaws barely parted, and the crushing dark pressing in from every side.

Where no photon from the surface has ever reached, scattered whale vertebrae lie half-buried in fine dark sediment, their bone-white surfaces flaring briefly into visibility each time a nearby organism emits a pulse of cyan-green bioluminescence — then vanishing again into near-total darkness. At pressures exceeding two hundred atmospheres, the water column above this carcass has become a vertical hunting ground, with viperfish hanging obliquely in the blackness, their needle teeth momentarily outlined by the cold living light of swarming scavengers, while the faint silhouette of a Magnapinna squid descends from upper darkness with filaments impossibly thin against the void. Anglerfish hover at different depths, their tiny lures emitting intermittent cyan pulses that ripple through clouds of marine snow drifting with absolute indifference through the cold — organic particles raining slowly downward, connecting the sunlit surface world to this lightless plain as they have for millions of years. A sulfide-darkened haze stains the sediment around the carcass edges where microbial mats process the last biochemical wealth of a life that ended far above, and far at the periphery of perception, a dim reddish-orange chemiluminescent glow suggests the presence of hydrothermal influence beyond the frame. Here the ocean exists entirely on its own terms: high-pressure, near-freezing, stratified by darkness and particle density, populated by predators whose entire evolutionary history is a negotiation with scarcity, silence, and the slow arithmetic of energy in the deep.

Between one thousand and four thousand meters below the surface, the midnight zone is a world of absolute cold, crushing pressure, and self-generated light. Here, where hydrostatic force reaches two hundred atmospheres or more, colonies of gelatinous medusae drift through vast black water in layered formation, their translucent bells and radial canals firing successive pulses of cold cyan and blue-green bioluminescence — a living canopy assembled not by any coordinated behavior but by the same dark currents that carry marine snow through the void in every direction. Each medusa is a pressure-adapted architecture of near-transparent mesoglea and delicate oral arms, tissue so gossamer it seems impossible under such compression, yet biochemically tuned to function perfectly in the cold and dark. Beneath this flickering ceiling of biological light, a deeper silhouette moves without illumination — a bathypelagic predator, perhaps a dragonfish or a large cephalopod, using the darkness itself as cover while scanning the luminous canopy above for the behavioral signatures of prey. This is the ocean as it has always been: layered, patient, and radically self-sufficient, its only light produced by the animals themselves, its only sound the faint hydrodynamic pressure of bodies passing through water that has never known the sun.

In the midnight zone between one and four kilometers down, where hydrostatic pressure exceeds two hundred atmospheres and cold hovers near two degrees Celsius, a solitary predator moves through open black water with the unhurried deliberateness of a creature shaped entirely by scarcity. Its maroon-black body, likely a dragonfish or elongate bathypelagic form, absorbs rather than reflects, rendering it nearly indistinguishable from the surrounding void except where sparse cyan photophores trace the briefest outline of its passage — bioluminescent organs that serve simultaneously as lure, camouflage-matching counterillumination, and species signal in a world where light is currency. All around it, marine snow descends in continuous silence: the slow, steady rain of organic detritus, fecal pellets, shed exoskeletons, and microbial aggregates that constitutes the only meaningful food link between the sunlit surface and the abyssal floor far below. The plain dimly legible beneath — flat, silted, basaltic — speaks to millions of years of particle accumulation over oceanic crust, its subtle relief fading without boundary into surrounding darkness, while at the horizon's edge a faint orange-red chemiluminescent haze suggests a hydrothermal plume too remote to warm or illuminate, only to remind that even here, energy finds its way. This ocean has no witness; it has always been exactly this — pressured, cold, immense, and entirely itself.
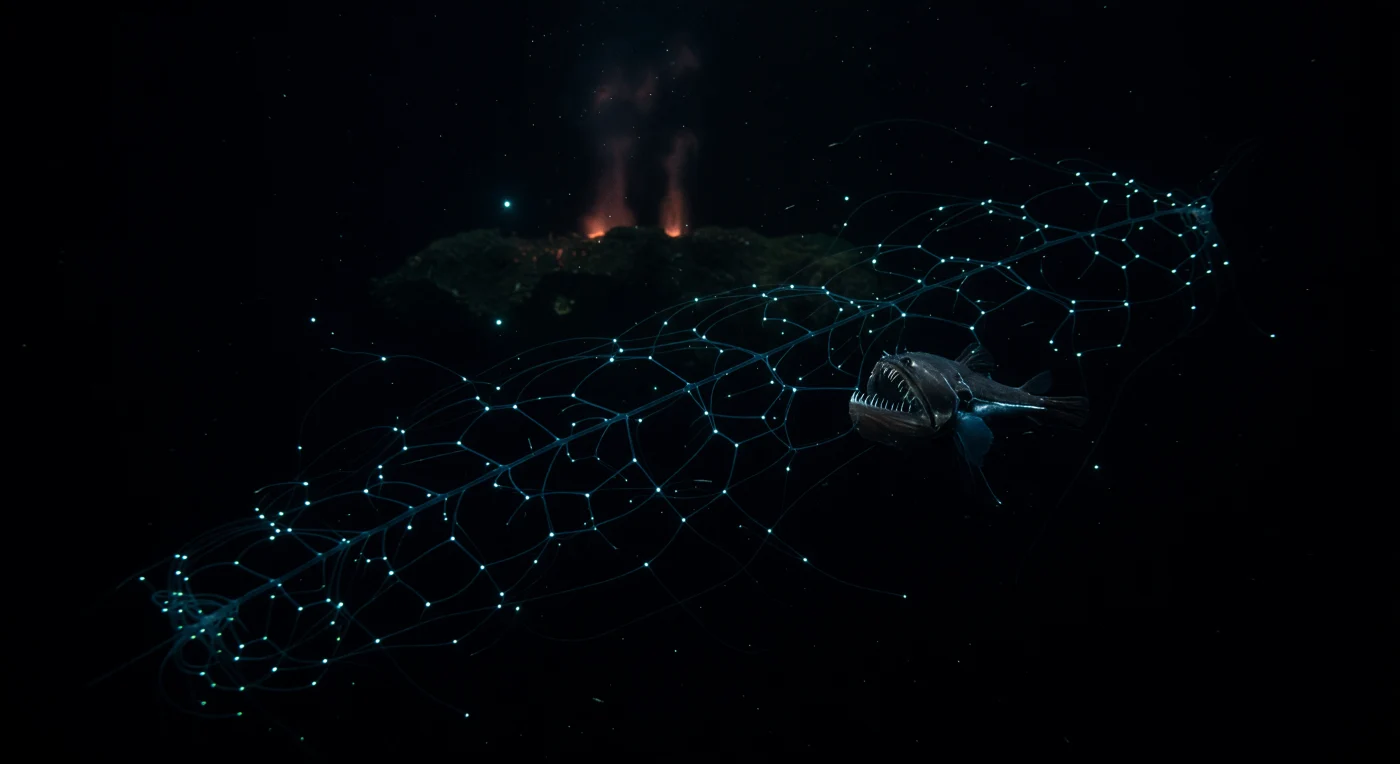
In the permanent darkness between one and four kilometers down, where pressure exceeds two hundred atmospheres and cold seawater hovers near two degrees Celsius, a siphonophore — not a single animal but a superorganism of coordinated zooids — suspends itself across the open water column like a living constellation, each nectophore and dactylozooid emitting soft blue-green photophores that collectively form a luminous geometric lattice, a passive trap evolved over millions of years to lure and snare whatever drifts within reach. Chauliodus sloani, the viperfish, positions itself just beyond this bioluminescent architecture in near-perfect ambush stillness, its elongated body barely distinguishable from the surrounding blackness, its needle-like fang teeth — proportionally the largest of any fish relative to head size — catching only intermittent cold cyan sparks from nearby zooplankton flashes and the siphonophore's own glow. Marine snow descends continuously through the water column in this zone, mineral particles and organic aggregates drifting like slow debris through a world where there is no sunlight, no season, and no surface rhythm, only the sporadic logic of bioluminescent deception and sudden predation. Far below in the background, a hydrothermal vent province exhales a faint chemiluminescent haze above fresh basalt, its reduced sulfur compounds supporting chemosynthetic communities entirely independent of photosynthesis, a reminder that energy in the deep ocean flows through pathways the sunlit world knows nothing of. This is a cold, pressurized, ancient darkness that functions with complete indifference to observation — a predator and its trap suspended in black water, waiting in a silence that has no need of witnesses.

At the fractured margin of a hydrothermal vent field somewhere between one and four kilometers beneath the surface, dense forests of giant tube worms — Riftia pachyptila and their relatives — rise from sulfide-encrusted basalt in tightly packed colonies, their ivory tubes topped with soft crimson plumes that sway in the slow convective currents rising from below, sustained not by sunlight but by the chemosynthetic metabolism of bacterial endosymbionts converting hydrogen sulfide into organic carbon. Black smoker plumes billow upward in layered mineral folds, and the restrained orange-red thermal glow seeping from fresh basalt seams, combined with the faint chemiluminescent haze of oxidizing mineral particulates, defines the only ambient light in water where photosynthetically active radiation has been functionally absent for hundreds of meters above. Along the crowded perimeter of the colony, small bioluminescent invertebrates pulse cyan and blue against wet chitin and sulfide-stained rock, and it is precisely here, at the lit edge of this chemosynthetic oasis, that an abyssal anglerfish holds itself motionless — its pressure-adapted body matte and dense, oversized jaws lined with translucent recurved teeth, its esca glowing cold greenish-cyan in a slow rhythmic pulse that mimics the very bioluminescence surrounding it, a lure evolved over millions of years to exploit the visual reflexes of prey that have no other light to follow. Beyond it, the vent field recedes into towering chimneys and smoky mineral veils, and somewhere deeper in the black water the needle silhouette of a viperfish and the improbable trailing filaments of a Magnapinna squid persist as barely resolved forms — pressure-tolerant bodies carrying the full evolutionary weight of an ecosystem that has never needed the sun.














